Today’s post is a review of some interesting new research that relates to the scope of this blog. It ranges from training young people with autism/ID to work at the car wash, to more complex science.
Let’s start with the easiest paper. Somewhat bizarrely it
was carried out in Japan by researchers from India. I am a fan of teaching kids
to wash cars but I was surprised to see that it would be covered in a published
research study.
One often forgotten item to teach teenagers and young adults
with autism or ID is how to safely use public transport, so they might travel
independently to and from any future job. We have had a lot of success with
this recently. Monty, now aged 20, can get all the way from home to various
different locations across the city using public transport, including changing
buses and with journey times more than one hour.
This study looked at how well visual task
evaluation helped teenagers with autism and intellectual disabilities become
more competent at car washing. For disabled people to promote their
independence and employment chances, car washing skills are crucial. The goal
of this study was to ascertain whether training techniques that include visual
task evaluation can improve car washing proficiency in teenagers with autism
and intellectual disabilities. 30 participants, ranging in age from 12 to 18,
participated in a pre-test/post-test design. Randomly chosen groups of
participants were put into the evaluation group for the visual task or the
control group. According to the findings, the visual task evaluation group
outperformed the control group in terms of car washing ability. Adolescents
with autism and intellectual disabilities can learn skills more quickly and
become more independent by including visual task evaluation into their teaching
strategies. These results demonstrate the potential for such treatments to enhance
their quality of life and employment chances.
Car washing
with a pressure washer is great fun for most people and washing a car thoroughly
has many individual steps to master, so it is good practice.
Head size
It has been known for decades that big heads (macrocephaly) and small heads (microcephaly) are a tell-tale sign of a neurodevelopment problem. Normally, big heads are linked to intellectual disability, but very small heads are also a warning sign.
Readers may recall the Zika virus epidemic in Brazil in 2015. This mosquito-borne virus caused pregnant women to give birth to children with microcephaly. Zika virus infection caused intellectual disability in babies. The severity of the intellectual disability varied from mild to severe. Babies with Zika virus infection may have difficulty learning and communicating. They may also have problems with problem-solving and abstract thinking. Hearing and vision can be impaired and growth is retarded.
Head size parts autism into two major subtypes
Essentially opposite paths in fetal brain development may explain two major subtypes of autism. In one of these subtypes, an unusually high number of excitatory neurons in a key brain region leads to large heads, or macrocephaly, which affects roughly 20 percent of people with autism; in the other, a decreased number of the same cells in that area leads to more typical head sizes, a new study finds.
This
fundamental biological difference suggests that “therapeutic avenues may be
drastically different for these subtypes,” says lead investigator Flora Vaccarino, professor of neuroscience
at Yale University. “That in turn could explain why drug treatments for autism
so far are failing.”
The opposite brain development paths found in this research may both lead to autism because they are each a case of imbalance, says investigator Alexej Abyzov, associate professor of biomedical informatics at the Mayo Clinic in Rochester, Minnesota.
The full paper:-
Idiopathic autism spectrum
disorder (ASD) is highly heterogeneous, and it remains unclear how convergent
biological processes in affected individuals may give rise to symptoms. Here,
using cortical organoids and single-cell transcriptomics, we modeled alterations
in the forebrain development between boys with idiopathic ASD and their
unaffected fathers in 13 families. Transcriptomic changes suggest that ASD
pathogenesis in macrocephalic and normocephalic probands involves an opposite
disruption of the balance between excitatory neurons of the dorsal cortical
plate and other lineages such as early-generated neurons from the putative
preplate. The imbalance stemmed from divergent expression of transcription
factors driving cell fate during early cortical development. While we did not
find genomic variants in probands that explained the observed transcriptomic
alterations, a significant overlap between altered transcripts and reported ASD
risk genes affected by rare variants suggests a degree of gene convergence between rare forms of ASD and the
developmental transcriptome in idiopathic ASD.
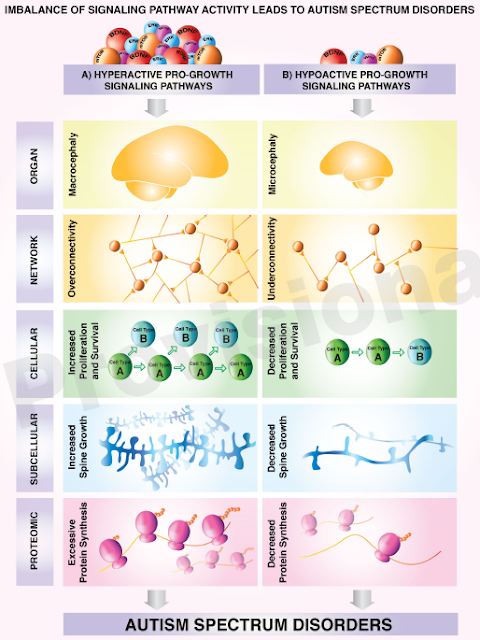
Head circumference at birth is a useful measurement, but what really matters is how it changes over time. Hyperactive pro-growth signaling affects more than just brain growth, it also affects muscle development, which is easy to notice. I have highlighted the graphic below several times in this blog and in my book. It is a good summary of what is going on.
Kv3.1
Regular readers will know that I like ion channels. The
reason is that dysfunctions in these channels really should be treatable. Usually we are looking for channel blockers,
but today with Kv3.1 we are looking for channel enhancers.
Ion channel enhancers increase the activity of ion channels
without directly opening them. They do this by increasing the number of open
channels, increasing the opening time of each channel, or decreasing the
closing time of each channel.
At the heart of the study is a type of inhibitory
neuron called GABAergic interneurons, which connect brain regions, playing
vital roles in coordinating high-frequency brain activity. As a potential source of the
excitatory/inhibitory imbalance in ASD and schizophrenia, evidence now points
to malfunction of a type of potassium channel, Kv3.1, special to GABAergic
interneurons. Denton
and his team will aim to develop Kv3.1 enhancers and test their efficacy in
restoring the balance of neural activity in a mouse model of ASD. In
latter stages of this work, they’ll focus on key brain areas, using various lab
techniques to carefully fill in neurological details surrounding any targeted
drug effects.
“This grant creates opportunities for developing
critically needed tool compounds to explore the role of Kv3.1 potassium
channels in autism spectrum disorder and schizophrenia,” said Denton, professor
of Anesthesiology and Pharmacology. “These are some of the most challenging and
costly disorders going, and we’re excited to have this opportunity to take this
work forward.”
Japanese researchers from the RIKEN Brain Science Institute are also thinking along the lines of targeting Kv3.1 to “correct aberrant developmental trajectories”.
Kv3.1 channels regulate the rate of critical period plasticity
The emergent function of fast-spiking PV-cell
circuits during postnatal life may hold the key to a deeper understanding of
critical periods in brain development (Reh et al.,
2020) and the etiology of related mental illnesses as
well (Do KQ and
Hensch, 2015). The human
neocortex notably shows a decrease in Kv3.1b channel protein
in schizophrenia, a deficit that is
restored by anti-psychotic drugs (Yanagi et
al., 2014). Moreover,
individuals with a KCNC1 loss-of-function
variant can present intellectual disability without seizure and epilepsy (Poirier et
al., 2017; Park et
al., 2019). Our work points toward a
prophylactic psychiatry that may target
these particular channels to correct aberrant developmental trajectories.
As with head size, the “when” is also important with
correcting Kv3.1. The idea is to intervene
at a very early age to redirect the developmental trajectory, rather than just to
improve today’s functioning.
The logical question is what drugs will Professor
Denton come up with to explore the benefit of targeting Kv3.1. Perhaps someone can beat him to it and save
us all a couple of decades?
If you look up Kv3.1 or the gene that encodes it
called KCNC1 you can read all about it.
https://www.genecards.org/cgi-bin/carddisp.pl?gene=KCNC1
As expected, there is no shortage of channel
blockers – Nifedipine (used a calcium channel blocker), Miconazole (an antifungal), Capsaicin (an
active component of chili peppers), Fluoxetine (better known as Prozac, which
is vitamin P to many people) plus many more.
Professor Denton is hunting for a channel enhancer. Keep an eye on what he comes up with. He has $2.7 million over 4 years to play with.
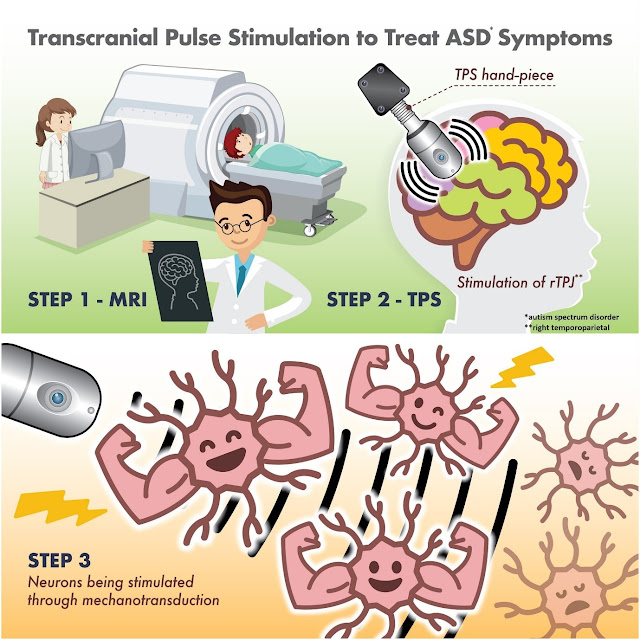
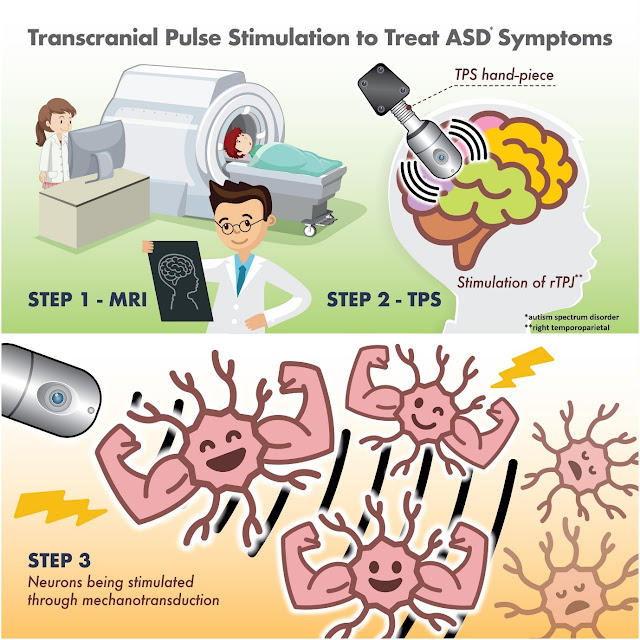
Transcranial pulse stimulation
Many autism parents do not like drug therapies, but
often like the idea of zapping the brain from outside. I liked the idea of Photo
biomodulation (PBMT) a form of light therapy that utilizes light sources
including lasers or LEDs.
Low
Level Laser Therapy (LLLT) for Autism – seems to work in Havana
Home/Clinic
based Photobiomodulation/Laser Therapy in Autism - acting on Light Sensitive
Ion Channels, Mitochondria, Lymph Nodes and more
You could potentially do Low Level Laser Therapy
(LLLT) at home.
Professor Manual Casanova is a fan of transcranial
magnetic stimulation (TMS).
Today’s paper below is about transcranial pulse
stimulation, which I suppose we can just call TPS.
Transcranial pulse stimulation (TPS) is a
non-invasive brain stimulation technique that uses pulsed electrical or
magnetic fields to stimulate the brain. It is a relatively new technique, but
it has the potential to be used for a variety of purposes, including:
- Treating neurological disorders such as Parkinson's disease, Alzheimer's disease, and depression
- Enhancing cognitive function, such as memory and attention
- Improving mood and well-being
- Reducing pain
- Promoting neuroplasticity, the ability of the brain to change and adapt
Transcranial pulse stimulation has been proven effective to
improve cognition, memory and depressive symptoms of Alzheimer’s disease, but
supporting evidence on other neurological diseases or neuropsychiatric
disorders remains limited. This study aimed to investigate the effects of
transcranial pulse stimulation on the right temporoparietal junction, which is
a key node for social cognition for autism spectrum disorder, and to examine
the association between transcranial pulse stimulation and executive and social
functions. This double-blinded, randomized, sham-controlled trial included 32
participants (27 males), aged 12–17 years with autism spectrum disorder. All
eligible participants were randomized into either the verum or sham
transcranial pulse stimulation group, on a 1:1 ratio, based on the Childhood
Autism Rating Scale screening score. Sixteen participants received six verum
transcranial pulse stimulation sessions (energy level: 0.2–0.25 mJ/mm2; pulse
frequency: 2.5–4.0 Hz, 800 pulse/session) in 2 weeks on alternate days. The
remaining 16 participants received sham transcranial pulse stimulation. The
primary outcome measure included Childhood Autism Rating Scale score changes,
evaluated by parents, from baseline to 3-month follow-ups. Secondary outcomes
included a self-reported questionnaire responded to by parents and cognitive
tests responded to by participants. A licensed mental health professional
evaluated clinical global impression severity, improvement, efficacy and total
score. Results revealed
significant interactions in Childhood Autism Rating Scale and other secondary
outcomes. Significant group and time effects were found in most
secondary outcomes. Additionally, significant differences were found between
the transcranial pulse stimulation and sham transcranial pulse stimulation
groups in Childhood Autism Rating Scale and clinical global impression
improvement and total score immediately after 2 weeks of transcranial pulse
stimulation intervention (all P < 0.05), and effects were sustainable at 1-
and 3-month follow-up, compared with baseline. The effect size of Childhood
Autism Rating Scale (d = 0.83–0.95) and clinical global impression improvement
(d = 4.12–4.37) were large to medium immediately after intervention and
sustained at 1-month post-stimulation; however, the effects were reduced to
small at 3-month post-stimulation (d = 2.31). These findings indicated that transcranial pulse
stimulation over right temporoparietal junction was effective to reduce the
core symptoms of autism spectrum disorder, as evidenced by a 24% reduction in
the total Childhood Autism Rating Scale score in the verum transcranial pulse
stimulation group. Additionally, the clinical global impression total
score was reduced by 53.7% in the verum transcranial pulse stimulation group at
a 3-month follow-up, compared with the baseline. Participants in the verum
transcranial pulse stimulation group had shown substantial improvement at 1-
and 3-month follow-ups, compared with baseline, although some of the
neuropsychological test results were deemed statistically insignificant. Future
replication of this study should include a larger sample derived from
multi-nations to determine transcranial pulse stimulation as an alternative
top-on treatment option in neuropsychiatry
TPS looks
pretty impressive, based on the above study. TPS is available today, but it
does need a lot of visits to the therapist. The effects are not permanent so
you would have to keep going back for more.
People are doing transcranial direct current stimulation (tDCS) at home.
People are zapping their brains at home to improve focus and clear brain fog. But is it safe?
For any kind
of zapping therapy to be viable, it would have to be possible to do it yourself
at home.
Targeting
alpha 5 subunit of GABAA
receptors
Some earlier
posts in this blog did get rather complicated.
One field that I looked at in rather painful detail was the GABAA
receptor. Some readers of this blog have children whose autism is entirely
caused by a defect in this receptor, many other readers just see the effects of
a GABAA malfunction caused by a problem with NKCC1/KCC2 expression resulting
from the GABA developmental switch failing to occur.
I looked to
me that targeting alpha 3 and alpha 5 subunits could well enhance cognition.
Alpha 3 is targeted
by low dose Clonazepam, thanks to Professor Catterall.
Alpha 5 was targeted
to treat Down syndrome, using a new drug called Basmisanil (an inverse agonist
of alpha 5 subunit of GABAA). That work failed. I wrote about Cardiazol/
Pentylenetetrazol (PTZ) a drug that was widely used in the 1930s in mental hospitals
to trigger seizures that were supposed to treat people with schizophrenia. At much lower doses, it found a new purpose
decades ago as an ingredient in cough medicine.
The alpha 5
subunit is one of several subunits that can make up a GABAA
receptor. GABAA receptors containing the alpha 5 subunit are thought
to be involved in cognitive function, learning and memory, and mood regulation.
PTZ has been shown to block the action of GABA at alpha 5-containing GABAa receptors in animal studies.
Variable Expression of GABRA5 and Activation of α5 - a Modifier of Cognitive Function in Autism?
Sodium Benzoate and GABRA5 - Raising Cognitive Function in Autism
Cardiazol, a failed Schizophrenia treatment from the 1930s,
repurposed at low doses as a Cognitive Enhancer in Down Syndrome and likely
some Autism
The logical human trial would be to use the cough mixture, Cardiazole that is already used in children.
“We actual have quite a few readers from India and that is
the only other country using this drug. In India the producer is Nicholas
Piramal and the brand name is Cardiazol Dicodid, it cost 30 US cents for
10ml. So for less than $1, or 70 rupees, you might have a few months of
cognitive enhancement, that is less than some people pay for 1 minute of ABA
therapy.
If a few drops
of this children’s cough medicine improves cognition please lets us all know.”
Back to
recent research on alpha 5 that caught my attention.
Autism Spectrum Disorders (ASD) are characterized by core behavioral symptoms in the domains of sociability, language/communication, and repetitive or stereotyped behaviors. Deficits in the prefrontal and hippocampal excitatory/inhibitory balance due to a functional loss of GABAergic interneurons are proposed to underlie these symptoms. Increasing the postsynaptic effects of GABA with compounds that selectively modulate GABAergic receptors could be a potential target for treating ASD symptoms. In addition, deficits in GABAergic interneurons have been linked to dopamine (DA) system dysregulation, and, despite conflicting evidence, abnormalities in the DA system activity may underly some ASD symptoms. Here, we investigated whether the positive allosteric modulator of α5-containing GABAA receptors (α5-GABAARs) SH-053-2’F-R-CH3 (10 mg/kg) attenuates behavioral abnormalities in a rat model for autism based on in utero VPA exposure. We also evaluated if animals exposed to VPA in utero present changes in the ventral tegmental area (VTA) DA system activity using in vivo electrophysiology and if SH-053-2’F-R-CH3 could attenuate these changes. In utero VPA exposure caused male and female rats to present increased repetitive behavior (self-grooming) in early adolescence and deficits in social interaction in adulthood. Male, but not female VPA rats, also presented deficits in recognition memory as adults. SH-053-2’F-R-CH3 attenuated the impairments in sociability and cognitive function in male VPA-exposed rats without attenuating the decreased social interaction in females. Male and female adult VPA-exposed rats also showed an increased VTA DA neuron population activity, which was not changed by SH-053-2’F-R-CH3. Despite sex differences, our findings indicate α5-GABAARs positive allosteric modulators may effectively attenuate some core ASD symptoms
Fine tuning alpha 5, perhaps you need more, perhaps less?
Neurobiology and Therapeutic
Potential of α5-GABA Type A Receptors
Despite being a genetically heterogeneous disorder, the potential utility for mechanism-based GABAAR pharmacologic treatment with ASDs is supported by shared pathologies both in patients and related mouse models.
PAM
α5 GABAAR Therapeutic Applications
Neurodevelopmental
Disorders
Mouse
models of neurodevelopmental disorders that present with insufficient
inhibitory tone show improvement with positive modulators of GABAAR signaling.
In the Scn1a+/− mouse model of Dravet syndrome, a severe childhood epileptic
encephalopathy syndrome with hyperactivity and autism behaviors, abnormal
social behaviors and fear memory deficits were rescued following treatment with
a benzodiazepine, clonazepam (Han et al., 2014). In an ASD mouse model with
reduced GABAAR-mediated inhibition, the BTBR T+tf/J mouse, the α2,3 and 5 PAM
L-838,417, improved deficits in social interaction, repetitive behaviors, and
spatial learning (Han et al., 2014).
Autism spectrum disorder (ASD), as a common
neurodevelopmental disorder that encompasses impairments in social
communication and interaction, as well as repetitive and restrictive behavior,
still awaits an effective treatment strategy. The involvement of GABAergic
neurotransmission, and especially a deficit of GABA A receptors that contain
the α5 subunits, were implicated in pathogenesis of ASD. Therefore, we tested MP‐III‐022, a
positive allosteric modulator (PAM) selective for α5GABAA receptors, in Wistar
rats prenatally exposed to valproic acid, as an animal model useful for
studying ASD. Postweaning rats of both sexes were treated for 7 days
with vehicle or MP‐III‐022 at two doses pharmacokinetically determined as
selective, and thereafter tested in a behavioral battery (social interaction
test, elevated plus maze, spontaneous locomotor activity, and standard and
reverse Morris water maze). Additional rats were used for establishing a primary neuronal
culture and performing calcium imaging, and determination of hippocampal mRNA
levels of GABRA5, NKCC1, and KCC2. MP‐III‐022 prevented impairments in
many parameters connected with social, repetitive and restrictive behavioral
domains. The lower and higher dose was more effective in males and females,
respectively. Intriguingly, MP‐III‐022 elicited certain changes in control
animals similar to those manifested in valproate animals themselves. Behavioral
results were mirrored in GABA switch and spontaneous neuronal activity,
assessed with calcium imaging, and also in expression changes of three genes
analyzed. Our data support a role of α5GABAA receptors in pathophysiology of
ASD, and suggest a potential application of selective PAMs in its treatment,
that needs to be researched in a sex‐specific manner. Lay Summary In rats
prenatally exposed to valproate as a model of autism, a modulator of α5GABAA
receptors ameliorated social, repetitive and restrictive impairments, and,
intriguingly, elicited certain autism‐like changes in control rats. Behavioral results were mirrored
in GABA switch and spontaneous neuronal activity, and partly in gene expression
changes. This shows a role of α5GABAA receptors in pathophysiology of ASD, and
a potential application of their selective modulators in its treatment.
Note the researchers actually know
about the GABA switch and so measured mRNA levels of NKCC1 and KCC2.
Note also that the lower dose of MP‐III‐022 was more effective in
males and the higher dose in females.
We even have
the recent associated PhD thesis from Anja Santrač:-
The influence of positive modulation of GABAA
receptors containing the alpha5 subunit on behavioral changes of mice and rats
in models of autistic disorders
The role of α5 GABAA
receptors in learning and memory is well known. Therefore, we decided to
examine the effect of the selective positive allosteric modulator (PAM)
MP-III-022 on learning and memory of healthy animals, as well as GABRA5
expression. After demonstrating the needed tolerability and potential
procognitive effects, the ligand would be used in an animal model of autism
spectrum disorders (ASD). ASD is a neurodevelopmental disorder that encompasses
impairments in social communication and interaction, as well as repetitive and
restrictive behavior, still without an effective treatment. In this context,
animal models that imitate specific disease’s symptoms are an excellent tool of
translational research. Some of the most frequently used models are BTBR T+
tf/J mouse strain (BTBR) and valproate prenatal model (VPA). Our experiments
have shown that the variability of α5GABAA receptors’ roles depends on its
level of expression and localization, on the type and protocol of cognitive tasks, the timing of testing and intensity
of pharmacological modulation. Obtained results proved potential beneficial
effects of MP-III-022 in cognitive tasks. The BTBR model failed to express
sufficient face validity, while VPA demonstrated adequate face validity and in
part construct validity. Thus, we decided to subacutely apply MP-III-022 to
juvenile VPA rats. In control animals, treatment led to GABRA5 decrease and to
impairments similar to ones seen in ASD, suggesting the possible role of this
receptor in the pathogenesis of the disease. Most importantly, our results demonstrated the
potential of α5 GABAA receptor PAMs in secondary prevention and treatment of
ASD, with the caveat that the drug development program would require
adaptations tailored to sex-specific differences revealed.
Good job
Anja. For our Serbian speaking readers, here is the link to her thesis:-
Perhaps we
should connect her with Professor Ben-Ari?
Conclusion
Fine tuning
alpha 5 subunits of GABAA receptors really should be followed up. I think you need both options - a little bit
more and a little bit less. It did not work for Roche in Down syndrome, but the
potential remains.
Kv3.1 is another focused target for research, that very likely will become actionable.
Transcranial pulse stimulation, like all the other zapping therapies, looks interesting, but it needs to be packaged in way that can actually be implemented every day at home.
In the meantime, at least getting
your kid to wash the car is something we can all do.